The U.S. Food and Drug Administration has declared a “most serious type” recall for certain Abbott Diabetes Care FreeStyle Libre 3 and FreeStyle Libre 3 Plus glucose monitor sensors, warning that continued use could cause serious injury or death.
According to the FDA’s announcement, these devices may provide incorrect low glucose readings that fall below actual blood sugar levels. If undetected over time, such inaccuracies risk dangerous treatment errors for diabetes patients—including excessive carbohydrate intake or delayed insulin administration—potentially leading to severe health complications, hospitalization, or fatalities.
As of January 7, 2026, Abbott Diabetes Care has reported 860 serious injuries and seven deaths associated with this issue stemming from the faulty sensors. The affected devices are continuous glucose monitoring systems designed for individuals aged four and older, intended to replace traditional blood glucose testing.
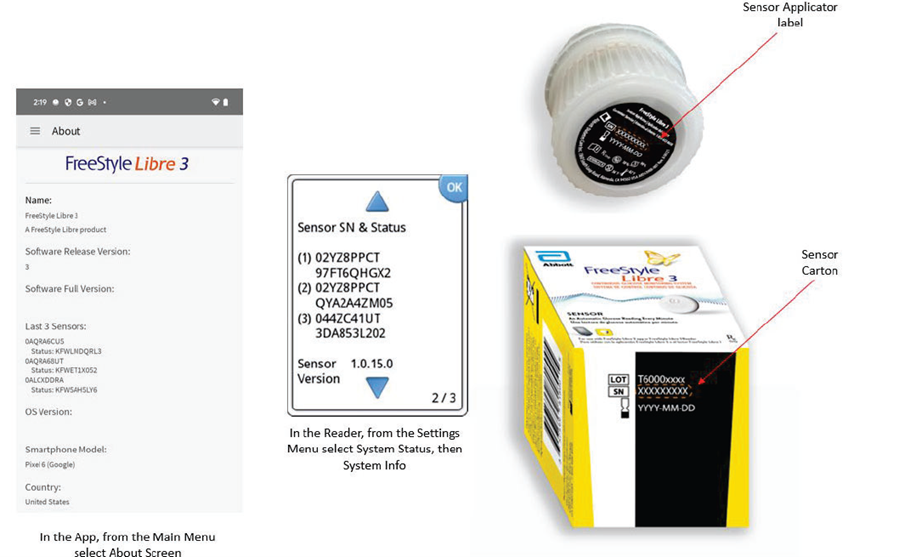
Patients can determine if their sensors are impacted by visiting www.freestylecheck.com and entering their sensor serial number—a detail found in the companion app, reader device, or on the sensor applicator label. Affected users must immediately stop using and dispose of the compromised devices.
Customers in the United States experiencing adverse reactions, quality concerns, or questions should contact Abbott Diabetes Care at 1-833-815-4273 or via www.freestyle.abbott/us-en/support/contact-us.html.